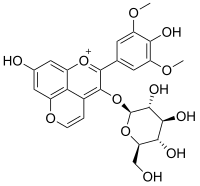
Vitisin B (piranoantocijanin)
Изглед
| |
| Nazivi | |
|---|---|
| Drugi nazivi
Piranomalvidin-3-glukozid
| |
| Identifikacija | |
3D model (Jmol)
|
|
| ChemSpider | |
| |
| Svojstva | |
| C25H25O12+ | |
| Molarna masa | 517,45 g/mol |
Ukoliko nije drugačije napomenuto, podaci se odnose na standardno stanje materijala (na 25 °C [77 °F], 100 kPa). | |
| Reference infokutije | |
Vitisin B je prirodni fenol prisutan u crvenim vinima.[1][2] It is a pyranoanthocyanin.[3][4][5]
Vidi još[уреди | уреди извор]
Reference[уреди | уреди извор]
- ^ Brazilian red wines made from the hybrid grape cultivar Isabel: Phenolic composition and antioxidant capacity. Suzana Lucy Nixdorf and Isidro Hermosín-Gutiérrez, Analytica Chimica Acta, Volume 659, Issues 1-2, 5 February 2010, Pages 208-215, Nixdorf, Suzana Lucy; Hermosín-Gutiérrez, Isidro (2010). „Brazilian red wines made from the hybrid grape cultivar Isabel: Phenolic composition and antioxidant capacity”. Analytica Chimica Acta. 659 (1–2): 208—215. PMID 20103126. doi:10.1016/j.aca.2009.11.058.
- ^ Morata, A.; Calderón, F.; González, M.C.; Gómez-Cordovés, M.C.; Suárez, J.A. (2007). „Formation of the highly stable pyranoanthocyanins (Vitisins a and B) in red wines by the addition of pyruvic acid and acetaldehyde”. Food Chemistry. 100 (3): 1144—1152. doi:10.1016/j.foodchem.2005.11.024.
- ^ Oliveira, Joana; De Freitas, Victor; Mateus, Nuno (2009). „A novel synthetic pathway to vitisin B compounds”. Tetrahedron Letters. 50 (27): 3933—3935. doi:10.1016/j.tetlet.2009.04.072.
- ^ Asenstorfer, Robert E.; Jones, Graham P. (2007). „Charge equilibria and pK values of 5-carboxypyranomalvidin-3-glucoside (Vitisin A) by electrophoresis and absorption spectroscopy”. Tetrahedron. 63 (22): 4788—4792. doi:10.1016/j.tet.2007.03.052.
- ^ Effect of acetaldehyde and several acids on the formation of vitisin A in model wine anthocyanin and colour evolution. Romero C. and Bakker J., International journal of food science & technology, 2000, vol. 35, no. 1. стр. 129-140, INIST:1283952
