Barijum-permanganat
Appearance
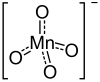
| |
| Identifikacija | |
|---|---|
3D model (Jmol)
|
|
| ECHA InfoCard | 100.029.191 |
| |
| |
| Svojstva | |
| BaMn2O8 | |
| Molarna masa | 375,198 |
Ukoliko nije drugačije napomenuto, podaci se odnose na standardno stanje materijala (na 25°C [77°F], 100 kPa). | |
| Reference infokutije | |
Barijum-permanganat je neorgansko hemijsko jedinjenje hemijske formule Ba(MnO4)2.
Dobijanje[uredi | uredi izvor]
Nastaje u reakciji barijum hlorida i srebro-permanganata.[3]
Upotreba[uredi | uredi izvor]
Koristi se za dobijanje permanganove kiseline.[3]
Osobine[uredi | uredi izvor]
| Osobina | Vrednost |
|---|---|
| Broj akceptora vodonika | 8 |
| Broj donora vodonika | 0 |
| Broj rotacionih veza | 0 |
| Particioni koeficijent[4] (ALogP) | -6,4 |
| Rastvorljivost[5] (logS, log(mol/L)) | 0,1 |
| Polarna površina[6] (PSA, Å2) | 238,8 |
Izvori[uredi | uredi izvor]
- ^ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today. 15 (23-24): 1052—7. PMID 20970519. doi:10.1016/j.drudis.2010.10.003.
- ^ Evan E. Bolton; Yanli Wang; Paul A. Thiessen; Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry. 4: 217—241. doi:10.1016/S1574-1400(08)00012-1.
- ^ a b Parkes, G.D. & Phil, D. 1973. Melorova moderna neorganska hemija. Naučna knjiga. Beograd.
- ^ Ghose, A.K.; Viswanadhan V.N. & Wendoloski, J.J. (1998). „Prediction of Hydrophobic (Lipophilic) Properties of Small Organic Molecules Using Fragment Methods: An Analysis of AlogP and CLogP Methods”. J. Phys. Chem. A. 102: 3762—3772. doi:10.1021/jp980230o.
- ^ Tetko IV, Tanchuk VY, Kasheva TN, Villa AE (2001). „Estimation of Aqueous Solubility of Chemical Compounds Using E-State Indices”. Chem Inf. Comput. Sci. 41: 1488—1493. PMID 11749573. doi:10.1021/ci000392t.
- ^ Ertl P.; Rohde B.; Selzer P. (2000). „Fast calculation of molecular polar surface area as a sum of fragment based contributions and its application to the prediction of drug transport properties”. J. Med. Chem. 43: 3714—3717. PMID 11020286. doi:10.1021/jm000942e.
Literatura[uredi | uredi izvor]
- Holleman A. F.; Wiberg E. (2001). Inorganic Chemistry (1st izd.). San Diego: Academic Press. ISBN 0-12-352651-5.
- Housecroft, C. E.; Sharpe, A. G. (2008). Inorganic Chemistry (3. izd.). Prentice Hall. ISBN 978-0-13-175553-6.
