Amiotrofična lateralna skleroza
| Amiotrofična lateralna skleroza (Lu Gerigova bolest) | |
|---|---|
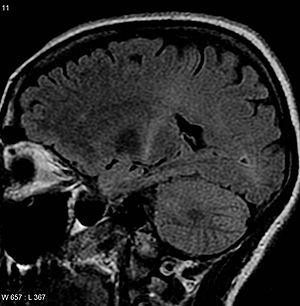 | |
| MRI (parasagittal FLAIR) demonstrira povišeni T2 signal u okviru zadnjeg dela unutrašnje kapsule, što je konsistentno sa kliničkom ALS dijagnozom | |
| Klasifikacija i spoljašnji resursi | |
| Specijalnost | neurologija |
| Patient UK | Amiotrofična lateralna skleroza |
Amiotrofična lateralna skleroza (ALS), Šarkoova bolest ili Lu Gerigova bolest (skraćeno ALS) pripada grupi neuroloških bolesti centralnog nervnog sistema, i primarno zahvata gornje i donje motorne neurone.[1][2][3] Jedna je od najčešćih bolesti prouzrokovanih degeneracijom motornih neurona. Ona može da se javi pojedinačno ili kao nasledna porodična bolest (10% svih slučajeva ALS-a). Za 20% nasleđenih ALS-a dokazana je mutacija gena za citosol[4][5], bakar/cink superoksid dismutazu (SOD 1).[6][7] Do danas je identifikovano gotovo 50 SOD 1 mutacija kod osoba s porodičnim ALS-om. Mada je svako podložan oboljenju, ALS najčešće pogađa sredovečne osobe.[8]
Čak 20.000 ljudi u SAD boluje od ALS-a, prema podacima Nacionalnog instituta za neurološke poremećaje i moždani udar, a svake godine dijagnostikuje se 5.000 novih slučajeva.
U početku ljudi mogu primetiti slabost u mišićima, trzanje ili grčenje, a potom bolest progresivno onesposobljava hod, govor, gutanje i na kraju disanje.[9] Mnogi bolesnici provedu poslednje dane života u potpunosti paralizovani, dok im mozak i dalje funkcioniše.[10]
Klinička slika je karakterisana progresivnim gubitkom mišićne mase i snage, fascikulacijama, spasticitetom, hiperrefleksijom i ekstenzornim plantarnim odgovorom (pozitivan znak Babinski refleksa), kao i razvojem dizartrije i disfagije.
Bolest ima brzi progresivni tok, a jedini odobreni lek od strane FDA je riluzol. Iako tretman riluzolom može produžiti život pacijenta, njegovi učinci su skromni.
Definicija[уреди | уреди извор]
| „ | Amiotrofična lateralna skleroza je bolest koja zahvata donji motorni neuron (DMN), lokalizovan u moždanom stablu i prednjim delovima kičmene moždine i prostire se do mišića i gornjeg motornog neurona (GMN), odnosno kortikospinalnog puta lokalizovanog u motornoj kori, koji se zatim prostire descendentno i inerviše donji motorni neuron.[11] | ” |
Istorija[уреди | уреди извор]

Prvi (nepotpuni) opis ove po mnoge bolesnike teske bolesti dao je Aran 1850. godine. Prema jednim 1869. ili po drugim izvorima 1873. godine, Žan-Marten Šarko (Jean-Martin Charcot; 1825—1893) , francuski neurolog, (koji se smatra osnivačem i prvim profesorom moderne neurologije), među prvima je opširnije opisao bolest i izdvojio ALS od drugih mišićnih atrofija. Naime on je uvideo da je kod ove bolesti mišićna atrofija udružena sa sklerozom bočnih snopova kičmene moždine. Njemu u čast bolest nosi naziv Šarkoova bolest, (Maladie de Charcot, Charcots disease). Ovaj naziv se najčešće koristi u francuskom govornom području.[12]
U Sjedinjenim Američkim Državama ALS je poznatija kao Lu Gerigova bolest (Lou Gehrig’s disease), po američkom igraču bejzbola obolelom od ove bolesti. Lu Gerig (Lou Gehrig) je bio američki bejzbol igrač koji je na vrhuncu svoje slave oboleo od ove bolesti i od nje umro 1941. godine. Tako se u Američkoj literaturi opis amiotrofične lateralne skleroze može često pronaći i pod njegovim imenom. Još jedno poznato ime je i Stiven Hoking, koji se smatra najistaknutijim astrofizičarem današnjice, koji je od bolesti motornog neurona bolovao od 1960. godine.[13]
Epidemiologija[уреди | уреди извор]
Incidenca i prevalenca ALS je oko 1 do 3 bolesnika na 100.000 novih slučajeva ili tri do pet bolesnika na 100.000 svih slučajeva godišnje.
- Morbiditet
U četiri regiona na zapadnom Pacifiku (Guam, Papua nova Gvineja, severna Australija i poluostrvo Ki u Japanu) posstoje endemska područja sa 5% do 10% familijarnih slučajeva ALS sa autozomno dominantnim tipom nasleđivanja, što je prvi put objavljeno 1993.god. u Britanskom časopisu „Nature“.
- Polne razlike
Kod radno produktivnih osoba mlađe životne dobi, muškarci češće obolevaju od žena, sa ukupnim muško-ženskim odnosom 1,5-2: 1 .[14]
U starijem životnom dobu, incidencija teži izjednačavanju polnih razlika, što se događa u dobi od 40 do 50 godine u nekim populacijama ili u dobi od 65 do 70 godina u drugima.[15]
- Životna dob
Većina obolelih je između 50 i 75 godina starosti. Napad ALS-a mogu se pojaviti od tinejdžerskih godina do kasnih 80-ih.
Incidencija raste s povećanjem dobi do starosti oko 75-80 godina. Srednja životna dob pojave sporadičnih ALS je 65 godina; prosečne dob za nastupanje familijarnih oblika ALS je u rasponu od 46 do 55 godina.[16]
- Smrtnost-mortalitet
ALS je progresivno, smrtonosno oboljenje, kod koga bolesnici pacijenti najčešče umiru od respiratorne paralize. Prosečno preživljavanje je tri do pet godina, mada se u literaturi navode slučajevi mnogo kraćeg ili mnogo dužeg preživljavanja.
Etiologija[уреди | уреди извор]
Bolesti motornog neurona (BMN) obuhvataju grupu oboljenja koja nastaju kao posledica oštećenja i nestajanja nervnih ćelija u mozgu i kičmenoj moždini koje su odgovorne za uspešno obavljanje pokreta i koordinaciju aktivnosti mišića. Te nervne ćelije zovu se motoneuroni. Ovakav tip oštećenja dovodi do funkcionalnih ispada i slabosti u mišićima ruku, nogu i mišićima koji su od značaja za disanje, govor i gutanje. Iz ove grupe bolesti motoneurona kod odraslih najučestalija je amiotrofična lateralna skleroza (ALS), kod koje su prisutni svi pomenuti simptomi.
Tačan mehanizam koji uzrokuje ALS nepoznat je za većinu sporadičnih ili sporadičnih porodičnih slučajeva. Kako je u većini slučajeva ALS sporadična, kauzalni uzroci ALS su najčešće sporadični, što otežava utvrđivanje etiologije i uzroka bolesti. Kod porodičnog oblika ALS kao mogući kauzalni uzročnik bolesti navode se mnogobrojne mutacije (anomalije) gena.
Mada je sklonost ka mutaciji gena primarno otkrivena kod porodičnog oblika ALS u pojedinim porodicama postoji i sklonost ka sporadičnim oblicima ALS. To je za očekivati, jer kao što postoji razlika između pojedinih članova unutar porodice tako se i pojava sporadičnih oblika bolesti zasniva na pravilno uzetoj porodičnoj istoriji bolesti, što opet zavisi o genskoj istoriji, veličini porodice, životnom dobu članova porodice, i nivoa znanja osobe od koje se uzima anamneza.[17]
Osim toga, bolesnici koji pripadaju prvom „kolenu“ ili prvostepenim srodnicima, sa naizgled sporadičnim oblikom bolesti, imaju povećan rizik od ALS. Opšti životni rizik od ALS kod ovih srodnika je međutim nizak (oko 1 u 50).[18]
Opšte informacije[уреди | уреди извор]
Kod ALS neurodegenerativni proces u motornim neuronima nalazi se u osnovi patoloških promena, u kojima dominira gubitak motornih ćelija prednjih rogova kičmene moždine i motornim jedrima donjega moždanog stabla.
- U prednjim rogovima kičmene moždine atrofiraju alfa motorne ćelije, dok su gama ćelije procesom degeneracije zahvaćene kasnije i u manjoj meri.
- U moždanom stablu oštećenja se najbolje vide u motornim jedrima X, XI. i XII. moždanog živca, manje u jedrima V. i VII moždanog živca. Uvek ostaju sačuvana tela pokretača očnih bulbusa i motorna jedra u sakralnim segmentima leđnog dela kičmene moždine koje inervišu vezikorektalne sfinktere. Iz brojnih istraživanja poznato je, da ovi neuroni nemaju adrenergičke receptore i zato su sličniji autonomnim neuronima. Vidljiva je asimetrija ovih lezija ogleda se i u nesimetričnosti kliničkih simptoma i znakova.[19]
- Degenerativne promene u citoplazmi
Neurofilamenti predstavljaju strukturalne belančevine u neuronu i ima ih više vrsta (NF-L, NF-H, NF-M). Količinom neurofilamenata određena je debljina nervnog vlakna. Oni su odgovorni su za aksoplazmatski transport, a promene u tom transportu uzrokuju različite patološke promene u neuronu. Fosforilirani neurofilamenti nakupljaju se u telu neurona i proksimalnom delu aksona u kojima se i razvija sfenoidna formacija koja se smatra početnom neurodegenerativnom promenom i najvjerojatnije uzrokom promene u aksonalnom transportu. Oticanje proksimalnih delova aksona verovatno prethodi vidljivim promenama u samim ćelijama. Smatra se da je akumulacija neurofilamenata glavni znak neurodegeneracije. Takve promene nalaze se kod porodičnog oblika ALS (PALS), sporadičnog oblika ALS (SALS), spinalnih mišićnih atrofija (SMA) i hereditarnih senzomotornih neuropatija (HSMN).[19]

Kao posledica promene neurofilamenata razvija se bolest motornog neurona, što je prvo utvrđeno kod eksperimentalnog miša. Na isti način promene neurofilamenata mogu nastati i kod čoveka. Karakteristične citoplazmatske inkluzije su posebna vrsta telašaca tzv. Bunina telešca koja su specifična za ALS. To su eozinofilne inkluzije veličine 2-7 mikrometara, koje se javljaju pojedinačno ili u grupicama stvarajući lance.
Kod ALS poznata je i pojava bazofilne inkluzije veličine 4-16 mikrometara, kao i obilnija pojava RNA koja se uočava kod bolesnika mlađih osoba. Poznati su i ubikutinski proteini, imunoreaktivne inkluzije, koje se stvaraju preko lizoma i specifične su samo za ALS.
Fragmentacija Goldžijevog aparata je rana patološka promena koja se uočava kod oko 30% bolesnika sa ALS. Telešca, kao što su Lewyjeva, su zapravo inkluzije koje se nalaze kod PALS kao i kod SALS. Ona su veličine 3-5 mikrometara i okružena su svetlim oreolom. Sadrže mikrotubule i vakuole a mogu biti udružena sa Buninim telašcima. U vidnom polju elektronskom mikroskopijom mogu se uočiti pahuljasti, nežni mikrotubuli i grubi endoplazmatski retikulum.
Opisane degenerativne promene postupno dovode do gubitka ćelija, a mnoge su od preživelih ćelija smanjene, „skupljene“, spongiozno izmenjene i ispunjene lipofuscinom. Propadanje ćelija u moždanoj kori i kičmenoj moždini nadoknađuje proliferacija glijalnih ćelija. Prednji koreni kičmene moždine su neuobičajeno tanki, a u njima postoji neproporcionalni gubitak velikih mijeliniziranih vlakana u motornim živcima. Posledično, u mišićnim vlaknima razvija se tipična denervacijskaa atrofija, različite starosti. Degeneracija i smanjenje prednjeg i bočnog piramidnog trakta je najjače izražena u cervikalnim (vratnim) i lumbalnim (slabinskim) segmentima kičmene moždine, ali se može pratiti i kroz moždano stablo sve do zadnjeg kraka kapsule interne i korone radijate.
Mikroskopski se vidi da su nestale Betzeove ćelije petog sloja precentralnog girusa motorne kore ali i prefrontalna motorne kore koja pokazuje degenerativne promene. To se može videti kao atrofija frontalnog režnja na MRT, a ponekad se i makroskopski vidi atrofija precentralnog girusa.
Degeneracija piramidnih puteva nije posledica atrofije moždane kore, jednako tako ni propadanje ćelija prednjih rogova nije izazvano transneuralnom degeneracijom, jer patološki proces može započeti na bilo kom nivou motoričkog sistema i u pojedinim presecima pokazuje oštećenjarazličitog stupena Piramidalni put je izmenjen često duž celog njegovog toka.[19]
Etiopatogeneza[уреди | уреди извор]

Na nivou ćelija postoji veliki broj mehanizama koji mogu pokrenuti proces degeneracije motornih neurona. Neki od tih mehanizama uključuju; mutaciju gena, oksidativni stres, toksičnost glutamata. U fazi istraživanja su i uticaj neurotrofičnih faktora, mogući poremećaj imunoloških funkcija, način života i faktori rizika iz spoljašnje sredine.[19]
- Neurotrofični faktori
U grupu faktora koji utiče na na normalan rast i regulaciju aktivnosti svih subpopulacija nervnih ćelija, spadaju faktori rasta nervne ćelije (nerve growth factor (NGF)). U neurotrofičke faktore odgovorne za razvoj motoneurona spadaju: neurotrophins, ciliary neurotrophic factor, hepatocyte growth factor, insulinlike growth factor, glialderived growth factor, itd.[22] Zato razvoj motoneurona zavisi od podrške neurotrofičnih faktora (npr. nedostatak glial growth faktora dovodi do redukcije Schwanovih ćelija i motoneurona.
Prema brojnim istraživanjima antiapoptotički signal neurotrofičnih faktora učestvuje u raznim patomehanizmima koji mogu biti odgovorni za smrt neurona. Tako ako je delovanje neurotrofičnih faktora neadekvatno dolazi do smrti motoneurona.
- Egzogeni neurotoksini
Na osnovu višegodišnjih istraživanja i brojnih epidemioloških studija o načinu života i uticaju spoljašnjih faktora koji bi mogli delovati kao „okidački mehanizmi“ na pojavu ALS, uočene su moguće veze obolelih osoba sa prethodnim izlaganjima mehaničkoj ili elektrotraumi, jonizujućem zračenju, virusnoj infekciji, raznim hemikalijama, teškim metalima (olovo, cink, živa, aluminijum ) i drugim elementima u tragovima. Nekoliko naučnih studija ustanovilo je statistički značajniju korelacije između ALS i izlaganja nekim poljoprivrednim pesticidima.[23][24][25][26]
Nedavno sprovedena studija o uticaju nepravilne ishrane ili dijetalne ishrane na rizik pojave bolesti otkrila je da su pacijenti sa ALS unosili u organizam manje vitamina E i nezasićenih masnoća u godini koja je prethodila razvoju simptoma. Međutim nije pouzdano dokazana direktna uzročna veza; pa se kao jedino moguće objašnjenje može prihvatiti stav da ti faktori, najverovatnije, samo povećavaju rizik za razvoja ALS, kumulativnim dejstvom ili slabljenjem funkcija nervnih ćelija, čineći ih osjetljivijima na procese degeneracije.[19]
Klinička slika[уреди | уреди извор]

Potpuno razvijenu kliničku sliku ALS karakteriše kombinacija atrofične i spastičke oduzetosti. Bolest je u načelu asimetrična. Ne postoje jasna pravila koja mogu objasniti raspodelu inicijalnih simptoma i njihovo dalje intenziviranje.
Kod otprilike 50% bolesnika ALS započinje atrofijama malih mišića šaka, zatim se javlja paraspastičnost nogu, da bi u daljem toku došlo do zahvatanja i područja inervacije motornih moždanih živaca.
Bolest često može započeti sa atrofičnim ili spastičnim parezama potkolenica i stopala, koje zatim zahvataju ruke i bulbarne mišiće (ascendentni oblik). U oko 20% bolesnika inicijalni simptomi su bulbarne pareze, koje se manifestuju smetnjama govora i gutanja (descendentni oblik).
Ako su prisutne centralne i periferne bulbarne smetnje često se kod takvih bolesnika javlja patološki (prisilni) smeh i plač. Osim toga kliničkom slikom mogu dominirati i mnogi drugih razvojni tipovi bolesti. Naročito često izostaju patološki refleksi. Fascikulacije se često uočavaju, i u mišićima koji nisu paretični.
Senzitivni poremećaji, koji nadmašuju povremene blaže parestezije, ili pak poremećaji kontrole mikcije ne pripadaju kliničkoj slici ALS-a. Retko se javljaju psihičke promene poput depresije ili drugih blažih kognitivnih smetnji.
U odmaklom periodu bolesti disanje postaje otežano zbog oštećenja interkostalnih mišića i dijafragme, pa je slabost respiratorne muskulature čest je uzrok smrti.
Tok kliničke slike bolesti je nezaustavljivo progresivan. Vrsta po kojoj se bolest razvija i dob u kojoj se pojavljuje ne pružaju pouzdane prognostičke elemente. Što se bolest ranije pojavi, može se očekivati duži period preživljavanja. Polovina bolesnika umire unutar dve godine a 90% unutar šest godina od početka bolesti (krajnji periodi preživljavanja su u rasponu od 6 mesec, minimalno do 20 godina maksimalno).[19]
Dijagnoza[уреди | уреди извор]
| Metode ispitivanja | Preporučene metode (obavezno) |
U pojedinim slučajevima (prema potrebi) |
|---|---|---|
| Krv |
|
|
| Mokraća |
| |
| Likvor |
| |
| Radiološka dijagnostika |
|
|
| Neurofiziološka dijagnostika |
|
Dijagnostički kriterijumi za ALS:[уреди | уреди извор]
| Kriterijumi | Znaci i simptomi |
|---|---|
| Pozitivni kriterijumu (prisutni znaci) |
|
| Odsutnost sledećih simptoma |
|
| Simptomi koji idu u prilog dijagnoze ALS |
|
Diferencijalna dijagnoza[уреди | уреди извор]
| Lokalizacija promena | Bolesti |
|---|---|
| Moždano stablo i kičmena moždina |
|
| Ćelije prednjih rogova kičmene moždine |
|
| Bolesti korena, pleksusa, živaca |
|
| Mozak |
|
| Neuromišićna sinapsa |
|
| Mišić |
|
| Sistemske bolesti |
|
Terapija[уреди | уреди извор]

Kako etiopatogeneza ALS još nije razjašnjena tako i specifično lečenje bolesti nije moguće, pa se u terapiji najčešće sprovode sledeći simptomatski i suportivni postupci:
- U ranim stadijumima, fizikalna terapija u svrhu održavanja pokretljivosti
- Smanjenje spastičnosti mišića, miotonolitičkim lekovima
- Kupiranje bulbarnih simptoma antiholinergiskim lekovima, u cilju smanjenja salivacije (lučenja pljuvačke)
- Kupiranje simptoma u bolesnika s mijastenijskom komponentom ALS, primenom malih doza mestinona
- Otklanjanje smetnji disanja, sve učestalijom primenom asistiranog disanja metodom neinvazivne ventilacije (NIV), ili u težim oblicima primenom traheotomije
- Otklanjanje smetnji otežanog gutanja i ishrane, nosnoželudačnom sondom, eventualno gastrostomom.[19]
Vidi još[уреди | уреди извор]
Izvori[уреди | уреди извор]
- ^ Мишић, Милан, ур. (2005). Енциклопедија Британика. А-Б. Београд: Народна књига : Политика. стр. 43. ISBN 86-331-2075-5.
- ^ Kwong LK, Uryu K, Trojanowski JQ, Lee VM (2008). „TDP-43 proteinopathies: neurodegenerative protein misfolding diseases without amyloidosis”. Neurosignals. 16 (1): 41—51. PMID 18097159. S2CID 36021142. doi:10.1159/000109758.
- ^ Kerman A, Liu HN, Croul S, Bilbao J, Rogaeva E, Zinman L, Robertson J, Chakrabartty A (2010). „Amyotrophic lateral sclerosis is a non-amyloid disease in which extensive misfolding of SOD1 is unique to the familial form”. Acta Neuropathol. 119 (3): 335—44. PMID 20111867. S2CID 22951808. doi:10.1007/s00401-010-0646-5.
- ^ Reaume A, Elliott J, Hoffman E, Kowall N, Ferrante R, Siwek D, Wilcox H, Flood D, Beal M, Brown R, Scott R, Snider W (1996). „Motor neurons in Cu/Zn superoxide dismutase-deficient mice develop normally but exhibit enhanced cell death after axonal injury”. Nat Genet. 13 (1): 43—7. PMID 8673102. S2CID 13070253. doi:10.1038/ng0596-43.
- ^ Bruijn L, Houseweart M, Kato S, Anderson K, Anderson S, Ohama E, Reaume A, Scott R, Cleveland D (1998). „Aggregation and motor neuron toxicity of an ALS-linked SOD1 mutant independent from wild-type SOD1”. Science. 281 (5384): 1851—4. Bibcode:1998Sci...281.1851B. PMID 9743498. doi:10.1126/science.281.5384.1851.
- ^ Conwit, Robin A. (2006). „Preventing familial ALS: A clinical trial may be feasible but is an efficacy trial warranted?”. Journal of the Neurological Sciences. 251 (1–2): 1—2. ISSN 0022-510X. PMID 17070848. S2CID 33105812. doi:10.1016/j.jns.2006.07.009.
- ^ Al-Chalabi Ammar, P. Nigel Leig Recent advances in amyotrophic lateral sclerosis Current Opinion in Neurology, 2000. vol. 13.4 ; 397–405.
- ^ ALS Clinic at the Penn State Milton S. Hershey Medical Center Pristupljeno 1. 6. 2017.
- ^ "Amyotrophic Lateral Sclerosis Fact Sheet" Архивирано на сајту Wayback Machine (12. октобар 2010) on National Institute of Neurological Disorders and Stroke
- ^ Phukan J, Pender NP, Hardiman O (2007). „Cognitive impairment in amyotrophic lateral sclerosis”. Lancet Neurol. 6 (11): 994—1003. PMID 17945153. S2CID 25786746. doi:10.1016/S1474-4422(07)70265-X.
- ^ Brown RH. Amyotrophic lateral sclerosis. MedLink-Neurobase third 2000 edition
- ^ Биографија Жан-Мартен Шаркоа (Jean-Martin Charcot) на whonamedit.com, Приступљено 6. јануар 2011.
- ^ Lacković Zdravko. Ekscitacijske aminokiseline. Neurotransmitori u zdravlju i bolesti; Medicinski fakultet Sveučilišta u Zagrebu, 1994; 185-214.
- ^ Armon C. Epidemiology of ALS/MND. In: Shaw P and Strong M, eds. „Motor Neuron Disorders”. Elsevier Sciences. 2003: 167—206..
- ^ Manjaly ZR, Scott KM, Abhinav K, Wijesekera L, Ganesalingam J, Goldstein LH,; et al. (октобар 2010). „The sex ratio in amyotrophic lateral sclerosis: A population based study”. Amyotroph Lateral Scler. 11 (5): 439—42..
- ^ Leigh PN, Swash M. Motor Neuron Disaese, Springer Verlag, London, 1995.
- ^ Al-Chalabi, A.; Lewis, C. M. (2011). „Modelling the effects of penetrance and family size on rates of sporadic and familial disease”. Human Heredity. 71 (4): 281—8. PMID 21846995. S2CID 3808240. doi:10.1159/000330167.
- ^ Hanby MF, Scott KM, Scotton W, Wijesekera L, Mole T, Ellis CE; et al. (2011). „The risk to relatives of patients with sporadic amyotrophic lateral sclerosis.”. Brain. Dec. 134: 3454—7..
- ^ а б в г д ђ е Poeck Klaus. Amiotrofična lateralna skleroza. Neurologija; Školska knjiga Zagreb, 2000; 457-459.
- ^ Grad, Leslie I.; Rouleau, Guy A.; Ravits, John; Cashman, Neil R. (август 2017). „Clinical Spectrum of Amyotrophic Lateral Sclerosis (ALS)”. Cold Spring Harbor Perspectives in Medicine. 7 (8): a024117. PMC 5538408
 . PMID 28003278. doi:10.1101/cshperspect.a024117..
. PMID 28003278. doi:10.1101/cshperspect.a024117..
- ^ Van Damme, Philip; Robberecht, Wim; Van Den Bosch, Ludo (2017). „Modelling amyotrophic lateral sclerosis: Progress and possibilities”. Disease Models & Mechanisms. 10 (5): 537—549. PMC 5451175
 . PMID 28468939. doi:10.1242/dmm.029058.
. PMID 28468939. doi:10.1242/dmm.029058.
- ^ Schulte-Herbruggen O, Braun A, Rochlitzer S, Jockers- Scherubl MC, Hellweg R. „Neurotrophic factors – a tool for therapeutic strategies in neurological, neuropsychia- tric and neuroimmunological diseases?”. Curr Med Chem. 14 (22): 2318—29..
- ^ Bonvicini, F.; Marcello, N.; Mandrioli, J.; Pietrini, V.; Vinceti, M. (2010). „Exposure to pesticides and risk of amyotrophic lateral sclerosis: A population-based case-control study”. Annali dell'istituto Superiore di Sanita. 46 (3): 284—7. PMID 20847462. doi:10.4415/ANN_10_03_10.
- ^ Malek, A. M.; Barchowsky, A.; Bowser, R.; Youk, A.; Talbott, E. O. (2012). „Pesticide exposure as a risk factor for amyotrophic lateral sclerosis: A meta-analysis of epidemiological studies: Pesticide exposure as a risk factor for ALS”. Environmental Research. 117: 112—119. PMID 22819005. doi:10.1016/j.envres.2012.06.007.]
- ^ Vinceti, M.; Bottecchi, I.; Fan, A.; Finkelstein, Y.; Mandrioli, J. (2012). „Are environmental exposures to selenium, heavy metals, and pesticides risk factors for amyotrophic lateral sclerosis?”. Reviews on Environmental Health. 27 (1): 19—41. PMID 22755265. S2CID 38486119. doi:10.1515/reveh-2012-0002.
- ^ Kamel, F.; Umbach, D. M.; Bedlack, R. S.; Richards, M.; Watson, M.; Alavanja, M. C.; Blair, A.; Hoppin, J. A.; Schmidt, S.; Sandler, D. P. (2012). „Pesticide exposure and amyotrophic lateral sclerosis”. Neurotoxicology. 33 (3): 457—62. PMC 3358481
 . PMID 22521219. doi:10.1016/j.neuro.2012.04.001.
. PMID 22521219. doi:10.1016/j.neuro.2012.04.001.
Literatura[уреди | уреди извор]
- Brooks BR, Miller RG, Swash M, Munsat TL (децембар 2000). „El Escorial revisited: revised criteria for the diagnosis of amyotrophic lateral sclerosis”. Amyotroph Lateral Scler Other Motor Neuron Disord. 1 (5): 293—9..
- [Guideline] Miller RG, Jackson CE, Kasarskis EJ et al. Practice Parameter update: The care of the patient with amyotrophic lateral sclerosis: Drug, nutritional, and respiratory therapies (an evidence-based review). „Report of the Quality Standards Subcommittee of the American Academy of Neurology”. Neurology. 73: 1218—1226. 2009..
- Miller RG, Jackson CE, Kasarskis EJ, et al. Practice parameter update: The care of the patient with amyotrophic lateral sclerosis: multidisciplinary care, symptom management, and cognitive/behavioral impairment (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 73 (15): 1227—1233. 2009-10-13. Недостаје или је празан параметар
|title=(помоћ). - Hardiman O, van den Berg LH, Kiernan MC (2011-10-11). „Clinical diagnosis and management of amyotrophic lateral sclerosis”. Nat Rev Neurol. 7 (11): 639—49..
- King SJ, Duke MM, O'Connor BA. „Living with amyotrophic lateral sclerosis/motor neurone disease (ALS/MND): decision-making about 'ongoing change and adaptation'”. J Clin Nurs. 18 (5): 745—54. март 2009..
- Eisen A (април 2009). „Amyotrophic lateral sclerosis: A 40-year personal perspective”. J Clin Neurosci. 16 (4): 505—12.. [Medline].
- Phukan J, Hardiman O (фебруар 2009). „The management of amyotrophic lateral sclerosis”. J Neurol. 256 (2): 176—86..
- Wijesekera LC, Leigh PN. Amyotrophic lateral sclerosis. Orphanet J Rare Dis. 2009 Feb 3. 4:3.
- Brooks BR. Managing amyotrophic lateral sclerosis: slowing disease progression and improving patient quality of life. Ann Neurol. 2009 Jan. 65 Suppl 1:S17-23.
- Al-Chalabi A, Fang F, Hanby MF, Leigh PN, Shaw CE, Ye W; et al. (децембар 2010). „An estimate of amyotrophic lateral sclerosis heritability using twin data”. J Neurol Neurosurg Psychiatry. 81 (12): 1324—6..
- Hudson AJ. Clinical Neurology. The motor neuron diseases and related disorders. 1996. Vol. 4: 11-14.
- Kanouchi T, Ohkubo T, Yokota T (јул 2012). „Can regional spreading of amyotrophic lateral sclerosis motor symptoms be explained by prion-like propagation?”. J Neurol Neurosurg Psychiatry. 83 (7): 739—45..
- Renton AE, Majounie E, Waite A, Simón-Sánchez J, Rollinson S, Gibbs JR, et al. „A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD”. Neuron. 72 (2): 257—68. 2011-10-20..
- DeJesus-Hernandez M, Mackenzie IR, Boeve BF, Boxer AL, Baker M, Rutherford NJ; et al. (2011-10-20). „Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS”. Neuron. 72 (2): 245—56.. [Medline]. [Full Text].
- Seeley WW, Crawford RK, Zhou J, Miller BL, Greicius MD (2009-04-16). „Neurodegenerative diseases target large-scale human brain networks”. Neuron. 62 (1): 42—52.. [Medline]. [Full Text].
- Armon C. What is ALS?. Bedlack RS, Mitsumoto H, Eds. Amyotrophic Lateral Sclerosis: A Patient Care Guide for Clinicians. New York: Demos Medical Publishing; 2012. 1-23.
- Ravits JM, La Spada AR (2009-09-08). „ALS motor phenotype heterogeneity, focality, and spread: deconstructing motor neuron degeneration”. Neurology. 73 (10): 805—11.. [Medline]. [Full Text].
- Armon C. Epidemiology of ALS/MND. Shaw P and Strong M, eds. Motor Neuron Disorders. Elsevier Sciences: 2003. 167-206.
- Armon C. ALS 1996 and Beyond: New Hopes and Challenges. A manual for patients, families and friends. Fourth Edition. California: Published by the LLU Department of Neurology, Loma Linda; 2007.
- Neary D, Snowden JS, Gustafson L; et al. (децембар 1998). „Frontotemporal lobar degeneration: a consensus on clinical diagnostic criteria”. Neurology. 51 (6): 1546—54..
- Clinical and neuropathological criteria for frontotemporal dementia (април 1994). „The Lund and Manchester Groups”. J Neurol Neurosurg Psychiatry. 57 (4): 416—8..
- Ravits JM, La Spada AR (2009-09-08). „ALS motor phenotype heterogeneity, focality, and spread: deconstructing motor neuron degeneration”. Neurology. 73 (10): 805—11.. [Medline].
- Wijesekera LC, Mathers S, Talman P, Galtrey C, Parkinson MH, Ganesalingam J; et al. (2009-03-24). „Natural history and clinical features of the flail arm and flail leg ALS variants”. Neurology. 72 (12): 1087—94..
- Almeida V, de Carvalho M, Scotto M, Pinto S, Pinto A, Ohana B; et al. (март 2013). „Primary lateral sclerosis: predicting functional outcome”. Amyotroph Lateral Scler Frontotemporal Degener. 14 (2): 141—5..
- Barber SC, Shaw PJ (2010-03-01). „Oxidative stress in ALS: key role in motor neuron injury and therapeutic target”. Free Radic Biol Med. 48 (5): 629—41..
- Strong MJ (новембар 2001). „Progress in clinical neurosciences: the evidence for ALS as a multisystems disorder of limited phenotypic expression”. Can J Neurol Sci. 28 (4): 283—98..
- Shaw PJ (август 2005). „Molecular and cellular pathways of neurodegeneration in motor neurone disease”. J Neurol Neurosurg Psychiatry. 76 (8): 1046—57..
- Rothstein JD. Current hypotheses for the underlying biology of amyotrophic lateral sclerosis. Ann Neurol. 2009 Jan. 65 Suppl 1:S3-9.
- Rosen DR, Siddique T, Patterson D, Figlewicz DA, Sapp P, Hentati A, et al. Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature. 362 (6415): 59—62. 1993-03-04. Недостаје или је празан параметар
|title=(помоћ). - Shaw PJ, Ince PG, Falkous G, Mantle D (октобар 1995). „Oxidative damage to protein in sporadic motor neuron disease spinal cord”. Ann Neurol. 38 (4): 691—5..
- Simpson EP, Henry YK, Henkel JS, Smith RG, Appel SH (2004-05-25). „Increased lipid peroxidation in sera of ALS patients: a potential biomarker of disease burden”. Neurology. 62 (10): 1758—65..
- Mitsumoto H, Santella RM, Liu X, Bogdanov M, Zipprich J, Wu HC; et al. (јун 2008). „Oxidative stress biomarkers in sporadic ALS”. Amyotroph Lateral Scler. 9 (3): 177—83..
- Bensimon G, Lacomblez L, Meininger V. A controlled trial of riluzole in amyotrophic lateral sclerosis. ALS/Riluzole Study Group. New England Journal of Medicine. 330 (9): 585—91. 1994-03-03. Недостаје или је празан параметар
|title=(помоћ). - Lacomblez L, Bensimon G, Leigh PN, Guillet P, Meininger V. Dose-ranging study of riluzole in amyotrophic lateral sclerosis. Amyotrophic Lateral Sclerosis/Riluzole Study Group II. Lancet. 347 (9013): 1425—31. 1996-05-25. Недостаје или је празан параметар
|title=(помоћ). [Medline]. - Neumann M, Sampathu DM, Kwong LK; et al. (2006-10-06). „Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis”. Science. 314 (5796): 130—3.. [Medline].
- Arai T, Hasegawa M, Akiyama H; et al. (2006-12-22). „TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis”. Biochem Biophys Res Commun. 351 (3): 602—11.. [Medline].
- Tan CF, Eguchi H, Tagawa A; et al. (мај 2007). „TDP-43 immunoreactivity in neuronal inclusions in familial amyotrophic lateral sclerosis with or without SOD1 gene mutation”. Acta Neuropathol. 113 (5): 535—42..
- Mackenzie IR, Bigio EH, Ince PG; et al. (мај 2007). „Pathological TDP-43 distinguishes sporadic amyotrophic lateral sclerosis from amyotrophic lateral sclerosis with SOD1 mutations”. Ann Neurol. 61 (5): 427—34..
- Sreedharan J, Blair IP, Tripathi VB; et al. (2008-03-21). „TDP-43 mutations in familial and sporadic amyotrophic lateral sclerosis”. Science. 319 (5870): 1668—72..
- Kabashi E, Valdmanis PN, Dion P; et al. (мај 2008). „TARDBP mutations in individuals with sporadic and familial amyotrophic lateral sclerosis”. Nat Genet. 40 (5): 572—4..
- Van Deerlin VM, Leverenz JB, Bekris LM; et al. (мај 2008). „TARDBP mutations in amyotrophic lateral sclerosis with TDP-43 neuropathology: a genetic and histopathological analysis”. Lancet Neurol. 7 (5): 409—16..
- Yokoseki A, Shiga A, Tan CF; et al. (април 2008). „TDP-43 mutation in familial amyotrophic lateral sclerosis”. Ann Neurol. 63 (4): 538—42..
- Rutherford NJ, Zhang YJ, Baker M, et al. Novel mutations in TARDBP (TDP-43) in patients with familial amyotrophic lateral sclerosis. PLoS Genet. 2008 Sep 19. 4(9):e1000193. [Medline].
- Del Bo R, Ghezzi S, Corti S, et al. TARDBP (TDP-43) sequence analysis in patients with familial and sporadic ALS: identification of two novel mutations. Eur J Neurol. 16 (6): 727—32. јун 2009. Недостаје или је празан параметар
|title=(помоћ). [Medline]. - Hasegawa M, Arai T, Akiyama H,; et al. (мај 2007). „TDP-43 is deposited in the Guam parkinsonism-dementia complex brains”. Brain. 130: 1386—94.. [Medline].
- Schwab C, Arai T, Hasegawa M, Akiyama H, Yu S, McGeer PL. TDP-43 pathology in familial British dementia. Acta Neuropathol. 2009 Mar 13. [Medline].
- Geser F, Martinez-Lage M, Robinson J; et al. (фебруар 2009). „Clinical and pathological continuum of multisystem TDP-43 proteinopathies”. Arch Neurol. 66 (2): 180—9.. [Medline].
- Kwiatkowski TJ Jr, Bosco DA, Leclerc AL, et al. Mutations in the FUS/TLS gene on chromosome 16 cause familial amyotrophic lateral sclerosis. Science. 323 (5918): 1205—8. 2009-02-27. Недостаје или је празан параметар
|title=(помоћ). [Medline]. - Vance C, Rogelj B, Hortobagyi T; et al. (2009-02-27). „Mutations in FUS, an RNA processing protein, cause familial amyotrophic lateral sclerosis type 6”. Science. 323 (5918): 1208—11.. [Medline].
- Simpson CL, Lemmens R, Miskiewicz K, Broom WJ, Hansen VK, van Vught PW, et al. Variants of the elongator protein 3 (ELP3) gene are associated with motor neuron degeneration. Hum Mol Genet. 18 (3): 472—81. 2009-02-01. Недостаје или је празан параметар
|title=(помоћ). - Padhi AK, Kumar H, Vasaikar SV, Jayaram B, Gomes J. Mechanisms of loss of functions of human angiogenin variants implicated in amyotrophic lateral sclerosis. PLoS One. 2012. 7(2):e32479.
- Armon C. An evidence-based medicine approach to the evaluation of the role of exogenous risk factors in sporadic amyotrophic lateral sclerosis. Neuroepidemiology (јануар 2011). „2003 Jul-Aug”. J Neuropathol Exp Neurol. 22 (4): 217—28..
- Armon C (септембар 2005). „Acquired nucleic acid changes may trigger sporadic amyotrophic lateral sclerosis”. Muscle Nerve. 32 (3): 373—7..
- Armon C. (2009). „Smoking may be considered an established risk factor for sporadic ALS”. Neurology November 17. 73 (20): 1693—1698..
- de Jong SW, Huisman MH, Sutedja NA, van der Kooi AJ, de Visser M, Schelhaas HJ; et al. (2012-08-01). „Smoking, alcohol consumption, and the risk of amyotrophic lateral sclerosis: a population-based study”. Am J Epidemiol. 176 (3): 233—9..
- Esclaire F, Kisby G, Spencer P, Milne J, Lesort M, Hugon J (јануар 1999). „The Guam cycad toxin methylazoxymethanol damages neuronal DNA and modulates tau mRNA expression and excitotoxicity”. Exp Neurol. 155 (1): 11—21..
- Kisby GE, Standley M, Park T, et al. Proteomic analysis of the genotoxicant methylazoxymethanol (MAM)-induced changes in the developing cerebellum. J Proteome Res. 5 (10): 2656—65. октобар 2006. Недостаје или је празан параметар
|title=(помоћ). - Ravits J, Paul P, Jorg C (2007-05-08). „Focality of upper and lower motor neuron degeneration at the clinical onset of ALS”. Neurology. 68 (19): 1571—5..
- Körner S, Kollewe K, Fahlbusch M, et al. „Onset and spreading patterns of upper and lower motor neuron symptoms in amyotrophic lateral sclerosis”. Muscle Nerve. 43 (5): 636—42. мај 2011..
- Armon C (2008-09-16). „From clues to mechanisms: understanding ALS initiation and spread”. Neurology. 71 (12): 872—3.. [Medline].
- Frank SA. Evolution in health and medicine Sackler colloquium: Somatic evolutionary genomics: mutations during development cause highly variable genetic mosaicism with risk of cancer and neurodegeneration. Proc Natl Acad Sci U S A. „2010 Jan 26”. 107 Suppl. 1: 1725—30..
- DeJesus-Hernandez M, Mackenzie IR, Boeve BF, Boxer AL, Baker M, Rutherford NJ; et al. (2011-10-20). „Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS”. Neuron. 72 (2): 245—56..
- Renton AE, Majounie E, Waite A, Simón-Sánchez J, Rollinson S, Gibbs JR, et al. „A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD”. Neuron. 72 (2): 257—68. 2011-10-20.. [Medline].
- Cooper-Knock J, Hewitt C, Highley JR, Brockington A, Milano A, Man S,; et al. (март 2012). „Clinico-pathological features in amyotrophic lateral sclerosis with expansions in C9ORF72”. Brain. 135: 751—64..
- Chiò A, Borghero G, Restagno G, Mora G, Drepper C, Traynor BJ, et al. „Clinical characteristics of patients with familial amyotrophic lateral sclerosis carrying the pathogenic GGGGCC hexanucleotide repeat expansion of C9ORF72”. Brain. 135: 784—93. март 2012..
- Byrne S, Elamin M, Bede P, Shatunov A, Walsh C, Corr B; et al. (март 2012). „Cognitive and clinical characteristics of patients with amyotrophic lateral sclerosis carrying a C9orf72 repeat expansion: a population-based cohort study”. Lancet Neurol. 11 (3): 232—40..
- Stewart H, Rutherford NJ, Briemberg H, Krieger C, Cashman N, Fabros M, et al. Clinical and pathological features of amyotrophic* lateral sclerosis caused by mutation in the C9ORF72 gene on chromosome 9p. Acta Neuropathol. 123 (3): 409—17. март 2012. Недостаје или је празан параметар
|title=(помоћ). - Wijesekera LC, Mathers S, Talman P, Galtrey C, Parkinson MH, Ganesalingam J; et al. (2009-03-24). „Natural history and clinical features of the flail arm and flail leg ALS variants”. Neurology. 72 (12): 1087—94..
- Al-Chalabi A, Lewis CM (2011). „Modelling the effects of penetrance and family size on rates of sporadic and familial disease”. Hum Hered. 71 (4): 281—8..
- Hanby MF, Scott KM, Scotton W, Wijesekera L, Mole T, Ellis CE,; et al. (децембар 2011). „The risk to relatives of patients with sporadic amyotrophic lateral sclerosis”. Brain. 134: 3454—7..
- Pasinelli P, Brown RH (септембар 2006). „Molecular biology of amyotrophic lateral sclerosis: insights from genetics”. Nat Rev Neurosci. 7 (9): 710—23..
- Fogh I, Rijsdijk F, Andersen PM, Sham PC, Knight J, Neale B; et al. (август 2007). „Age at onset in sod1-mediated amyotrophic lateral sclerosis shows familiality”. Neurogenetics. 8 (3): 235—6..
- Ahmeti KB, Ajroud-Driss S, Al-Chalabi A, Andersen PM, Armstrong J, et al. The Algen Consortium. Age of onset of amyotrophic lateral sclerosis is modulated by a locus on 1p34.1. Neurobiol Aging. 2013 Jan. 34(1):357.e7-19.
- Andersen PM, Al-Chalabi A (2011-10-11). „Clinical genetics of amyotrophic lateral sclerosis: what do we really know?”. Nat Rev Neurol. 7 (11): 603—15..
- Ferraiuolo L, Kirby J, Grierson AJ, Sendtner M, Shaw PJ (новембар 2011). „Molecular pathways of motor neuron injury in amyotrophic lateral sclerosis”. Nat Rev Neurol. 7 (11): 616—30..
- Wu CH, Fallini C, Ticozzi N, Keagle PJ, Sapp PC, Piotrowska K; et al. (2012-08-23). „Mutations in the profilin 1 gene cause familial amyotrophic lateral sclerosis”. Nature. 488 (7412): 499—503..
- Andersen PM. Amyotrophic lateral sclerosis genetics with Mendelian inheritance. Brown RH Jr, Swash M and Pasinelli P, eds. Amyotrophic Lateral Sclerosis. 2nd edition. Informa healthcare; 2006. 187-207.
- Saeed M, Yang Y, Deng HX; et al. (2009-05-12). „Age and founder effect of SOD1 A4V mutation causing ALS”. Neurology. 72 (19): 1634—9..
- Saito Y, Yokota T, Mitani T; et al. (2005-12-30). „Transgenic small interfering RNA halts amyotrophic lateral sclerosis in a mouse model”. J Biol Chem. 280 (52): 42826—30..
- Al-Chalabi A, Jones A, Troakes C, King A, Al-Sarraj S, van den Berg LH (септембар 2012). „The genetics and neuropathology of amyotrophic lateral sclerosis”. Acta Neuropathol. 124 (3): 339—52..
- Arai T, Mackenzie IR, Hasegawa M, et al. Phosphorylated TDP-43 in Alzheimer's disease and dementia with Lewy bodies. Acta Neuropathol. 117 (2): 125—36. фебруар 2009. Недостаје или је празан параметар
|title=(помоћ). - Morita M, Al-Chalabi A, Andersen PM, Hosler B, Sapp P, Englund E; et al. (2006-03-28). „A locus on chromosome 9p confers susceptibility to ALS and frontotemporal dementia”. Neurology. 66 (6): 839—44..
- Vance C, Al-Chalabi A, Ruddy D, Smith BN, Hu X, Sreedharan J, et al. Familial amyotrophic lateral sclerosis with frontotemporal dementia is linked to a locus on chromosome 9p13.2-21.3. Brain. 129: 868—76. април 2006. Недостаје или је празан параметар
|title=(помоћ). - van Es MA, Veldink JH, Saris CG, Blauw HM, van Vught PW, Birve A, et al. Genome-wide association study identifies 19p13.3 (UNC13A) and 9p21.2 as susceptibility loci for sporadic amyotrophic lateral sclerosis. Nat Genet. 41 (10): 1083—7. октобар 2009. Недостаје или је празан параметар
|title=(помоћ). . - Shatunov A, Mok K, Newhouse S, Weale ME, Smith B, Vance C (октобар 2010). „Chromosome 9p21 in sporadic amyotrophic lateral sclerosis in the UK and seven other countries: a genome-wide association study”. Lancet Neurol. 9 (10): 986—94..
- Laaksovirta H, Peuralinna T, Schymick JC, Scholz SW, Lai SL, Myllykangas L (октобар 2010). „Chromosome 9p21 in amyotrophic lateral sclerosis in Finland: a genome-wide association study”. Lancet Neurol. 9 (10): 978—85..
- Smith BN, Newhouse S, Shatunov A, Vance C, Topp S, Johnson L, et al. The C9ORF72 expansion mutation is a common cause of ALS+/-FTD in Europe and has a single founder. Eur J Hum Genet. 21 (1): 102—8. јануар 2013. Недостаје или је празан параметар
|title=(помоћ). - Al-Sarraj S, King A, Troakes C, Smith B, Maekawa S, Bodi I, et al. p62 positive, TDP-43 negative, neuronal cytoplasmic and intranuclear inclusions in the cerebellum and hippocampus define the pathology of C9orf72-linked FTLD and MND/ALS. Acta Neuropathol. 122 (6): 691—702. децембар 2011. Недостаје или је празан параметар
|title=(помоћ). - Deng HX, Chen W, Hong ST, Boycott KM, Gorrie GH, Siddique N, et al. Mutations in UBQLN2 cause dominant X-linked juvenile and adult-onset ALS and ALS/dementia. Nature. 477 (7363): 211—5. 2011-08-21. Недостаје или је празан параметар
|title=(помоћ). - Majoor-Krakauer D, Ottman R, Johnson WG, Rowland LP. „Familial aggregation of amyotrophic lateral sclerosis, dementia, and Parkinson's disease: evidence of shared genetic susceptibility”. Neurology. 44 (10): 1872—7. октобар 1994..
- Fallis BA, Hardiman O (април 2009). „Aggregation of neurodegenerative disease in ALS kindreds”. Amyotroph Lateral Scler. 10 (2): 95—8..
- Cruz DC, Nelson LM, McGuire V, Longstreth WT Jr (1999). „Physical trauma and family history of neurodegenerative diseases in amyotrophic lateral sclerosis: a population-based case-control study”. Neuroepidemiology. 18 (2): 101—10..
- Nelson LM, McGuire V, Longstreth WT Jr, Matkin C. Population-based case-control study of amyotrophic lateral sclerosis in western Washington State. I (2000-01-15). „Cigarette smoking and alcohol consumption”. Am J Epidemiol. 151 (2): 156—63..
- Kamel F, Umbach DM, Munsat TL, Shefner JM, Sandler DP (1999). „Association of cigarette smoking with amyotrophic lateral sclerosis”. Neuroepidemiology. 18 (4): 194—202..
- Gallo V, Bueno-De-Mesquita HB, Vermeulen R, Andersen PM, Kyrozis A, Linseisen J (април 2009). „Smoking and risk for amyotrophic lateral sclerosis: analysis of the EPIC cohort”. Ann Neurol. 65 (4): 378—85..
- Sutedja NA, Veldink JH, Fischer K, Kromhout H, Wokke JH, Huisman MH (2007-10-09). „Lifetime occupation, education, smoking, and risk of ALS”. Neurology. 69 (15): 1508—14..
- Weisskopf MG, O'Reilly EJ, McCullough ML; et al. (2005-01-11). „Prospective study of military service and mortality from ALS”. Neurology. 64 (1): 32—7..
- Horner RD, Kamins KG, Feussner JR; et al. (2003-09-23). „Occurrence of amyotrophic lateral sclerosis among Gulf War veterans”. Neurology. 61 (6): 742—9.. [Medline].
- Armon C. Occurrence of amyotrophic lateral sclerosis among Gulf War veterans. Neurology. 2004 Mar 23. 62(6):1027; author reply 1027-9. [Medline].
- Armon C (2004-11-23). „Excess incidence of ALS in young Gulf War veterans”. Neurology. 63 (10): 1986—7.; author reply 1986-7.
- Coffman CJ, Horner RD, Grambow SC, Lindquist J. „Estimating the occurrence of amyotrophic lateral sclerosis among Gulf War (1990-1991) veterans using capture-recapture methods”. Neuroepidemiology. 24 (3): 141—50. 2005..
- Horner RD, Grambow SC, Coffman CJ; et al. (2008). „Amyotrophic lateral sclerosis among 1991 Gulf War veterans: evidence for a time-limited outbreak”. Neuroepidemiology. 31 (1): 28—32..
- Barth SK, Kang HK, Bullman TA, Wallin MT. Neurological mortality among U.S (септембар 2009). „veterans of the Persian Gulf War: 13-year follow-up”. Am J Ind Med. 52 (9): 663—70..
- Sutedja NA, Veldink JH, Fischer K, Kromhout H, Heederik D, Huisman MH, et al. Exposure to chemicals and metals and risk of amyotrophic lateral sclerosis: a systematic review. Amyotroph Lateral Scler. 2009 Oct-Dec. 10(5-6):302-9.
- Popat RA, Van Den Eeden SK, Tanner CM; et al. (2006). „Effect of reproductive factors and postmenopausal hormone use on the risk of amyotrophic lateral sclerosis”. Neuroepidemiology. 27 (3): 117—21..
- Beghi E, Logroscino G, Chiò A, Hardiman O, Millul A, Mitchell D, et al. „Amyotrophic lateral sclerosis, physical exercise, trauma and sports: results of a population-based pilot case-control study”. Amyotroph Lateral Scler. 11 (3): 289—92. 2010-05-03..
- Chiò A, Benzi G, Dossena M, Mutani R, Mora G. „Severely increased risk of amyotrophic lateral sclerosis among Italian professional football players”. Brain. 128: 472—6. март 2005..
- Belli S, Vanacore N (2005). „Proportionate mortality of Italian soccer players: is amyotrophic lateral sclerosis an occupational disease?”. Eur J Epidemiol. 20 (3): 237—42.. [Medline].
- Armon C. Sports and trauma in amyotrophic lateral sclerosis revisited. J Neurol Sci. 2007 Nov 15. 262(1-2):45-53. [Medline].
- Chio A, Traynor BJ, Swingler R, eet al. Amyotrophic lateral sclerosis and soccer: a different epidemiological approach strengthen the previous findings. J Neurol Sci. 2008 Jun 15. 269(1-2):187-8; author reply 188-9.
- Belli S, Vanacore N. Sports and amyotrophic lateral sclerosis. J Neurol Sci. 2008 Jun 15. 269(1-2):191; author reply 191-2.
- Armon C (2008). J Neurol Sci. 269: 188—189. Недостаје или је празан параметар
|title=(помоћ). - Armon C. Response to Belli and Vanacore. J Neurol Sci. 2008. 269:191192 (author reply).
- Chio A, Calvo A, Dossena M, Ghiglione P, Mutani R, Mora G (август 2009). „ALS in Italian professional soccer players: the risk is still present and could be soccer-specific”. Amyotroph Lateral Scler. 10 (4): 205—9..
- McKee AC, Gavett BE, Stern RA, Nowinski CJ, Cantu RC, Kowall NW; et al. (септембар 2010). „TDP-43 proteinopathy and motor neuron disease in chronic traumatic encephalopathy”. J Neuropathol Exp Neurol. 69 (9): 918—29..
- Appel SH, Cwik VA, Day JW (децембар 2010). „Trauma, TDP-43, and amyotrophic lateral sclerosis”. Muscle Nerve. 42 (6): 851—2.. [Medline].
- J Neuropathol Exp Neurol. 70 (1): 96—7. јануар 2011. Недостаје или је празан параметар
|title=(помоћ); author reply 98-100. [Medline]. - Armon C, Miller RG (2010). „Correspondence regarding: TDP-43 proteinopathy and motor neuron disease in chronic traumatic encephalopathy”. J Neuropathol Exp Neurol. 69: 918—29.; author reply 98-100.
- Lehman EJ, Hein MJ, Baron SL, Gersic CM (2012-11-06). „Neurodegenerative causes of death among retired National Football League players”. Neurology. 79 (19): 1970—4..
- Armon C, Nelson LM (јун 2012). „Is head trauma a risk factor for amyotrophic lateral sclerosis? An evidence based review”. Amyotroph Lateral Scler. 13 (4): 351—6..
- Peters TL, Fang F, Weibull CE, Sandler DP, Kamel F, Ye W (мај 2013). „Severe head injury and amyotrophic lateral sclerosis”. Amyotroph Lateral Scler Frontotemporal Degener. 14 (4): 267—72..
- Kurland LT, Radhakrishnan K, Smith GE, Armon C, Nemetz PN (децембар 1992). „Mechanical trauma as a risk factor in classic amyotrophic lateral sclerosis: lack of epidemiologic evidence”. J Neurol Sci. 113 (2): 133—43..
- Savica R, Parisi JE, Wold LE, Josephs KA, Ahlskog JE (2012). Mayo Clin Proc. 87: 335—340. Недостаје или је празан параметар
|title=(помоћ). - Wicks P, Ganesalingham J, Collin C, Prevett M, Leigh NP, Al-Chalabi A (јун 2007). „Three soccer playing friends with simultaneous amyotrophic lateral sclerosis”. Amyotroph Lateral Scler. 8 (3): 177—9.. [Medline].
- Whiting MG (1963). Economic Botany. 68: 270—302. Недостаје или је празан параметар
|title=(помоћ). - Borenstein AR, Mortimer JA, Schellenberg GD, Galasko D. The ALS/PDC syndrome of Guam and the cycad hypothesis. Neurology. 2009 Feb 3. 72(5):473, 476; author reply 475-6.
- Steele JC, McGeer PL. The ALS/PDC syndrome of Guam and the cycad hypothesis. Neurology. 70 (21): 1984—90. 2008-05-20. Недостаје или је празан параметар
|title=(помоћ). [Medline]. - Borenstein AR, Mortimer JA, Schofield E; et al. (2007-05-22). „Cycad exposure and risk of dementia, MCI, and PDC in the Chamorro population of Guam”. Neurology. 68 (21): 1764—71..
- Cox PA, Banack SA, Murch SJ (2003-11-11). „Biomagnification of cyanobacterial neurotoxins and neurodegenerative disease among the Chamorro people of Guam”. Proc Natl Acad Sci U S A. 100 (23): 13380—3..
- Khabazian I (2002). J Neurochem. 82: 516—528. Недостаје или је празан параметар
|title=(помоћ). - Chiò A, Mora G, Calvo A, Mazzini L, Bottacchi E, Mutani R. „Epidemiology of ALS in Italy: a 10-year prospective population-based study”. Neurology. 72 (8): 725—31. 2009-02-24..
- Alonso A, Logroscino G, Jick SS, Hernán MA. „Incidence and lifetime risk of motor neuron disease in the United Kingdom: a population-based study”. Eur J Neurol. 16 (6): 745—51. јун 2009..
- Johnston CA, Stanton BR, Turner MR, Gray R, Blunt AH, Butt D; et al. (децембар 2006). „Amyotrophic lateral sclerosis in an urban setting: a population based study of inner city London”. J Neurol. 253 (12): 1642—3..
- Johnston CA, Stanton BR, Turner MR, Gray R, Blunt AH, Butt D; et al. (децембар 2006). „Amyotrophic lateral sclerosis in an urban setting: a population based study of inner city London”. J Neurol. 253 (12): 1642—3..
- Cronin S, Hardiman O, Traynor BJ (2007-03-27). „Ethnic variation in the incidence of ALS: a systematic review”. Neurology. 68 (13): 1002—7..
- Zaldivar T, Gutierrez J, Lara G, Carbonara M, Logroscino G, Hardiman O (2009-05-12). „Reduced frequency of ALS in an ethnically mixed population: a population-based mortality study”. Neurology. 72 (19): 1640—5..
- Rojas-Garcia R, Scott KM, Roche JC, Scotton W, Martin N, Janssen A; et al. (јануар 2012). „No evidence for a large difference in ALS frequency in populations of African and European origin: a population based study in inner city London”. Amyotroph Lateral Scler. 13 (1): 66—8..
- Sabel CE, Boyle PJ, Löytönen M, et al. „Spatial clustering of amyotrophic lateral sclerosis in Finland at place of birth and place of death”. Am J Epidemiol. 157 (10): 898—905. 2003-05-15..
- Manjaly ZR, Scott KM, Abhinav K, Wijesekera L, Ganesalingam J, Goldstein LH; et al. (октобар 2010). „The sex ratio in amyotrophic lateral sclerosis: A population based study”. Amyotroph Lateral Scler. 11 (5): 439—42..
- Kimura F, Fujimura C, Ishida S; et al. (2006-01-24). „Progression rate of ALSFRS-R at time of diagnosis predicts survival time in ALS”. Neurology. 66 (2): 265—7..
- Turner MR, Scaber J, Goodfellow JA, Lord ME, Marsden R, Talbot K. The diagnostic pathway and prognosis in bulbar-onset amyotrophic lateral sclerosis. J Neurol Sci. 2010 Jul 15. 294(1-2):81-5.
- Murphy J, Henry R, Lomen-Hoerth C (март 2007). „Establishing subtypes of the continuum of frontal lobe impairment in amyotrophic lateral sclerosis”. Arch Neurol. 64 (3): 330—4..
- Phukan J, Elamin M, Bede P, Jordan N, Gallagher L, Byrne S; et al. (јануар 2012). „The syndrome of cognitive impairment in amyotrophic lateral sclerosis: a population-based study”. J Neurol Neurosurg Psychiatry. 83 (1): 102—8..
- Elamin M, Phukan J, Bede P, Jordan N, Byrne S, Pender N; et al. (2011-04-05). „Executive dysfunction is a negative prognostic indicator in patients with ALS without dementia”. Neurology. 76 (14): 1263—9..
- Chiò A, Logroscino G, Hardiman O, Swingler R, Mitchell D, Beghi E, et al. Prognostic factors in ALS: A critical review. Amyotroph Lateral Scler. 2009 Oct-Dec. 10(5-6):310-23.
- Kimura F, Fujimura C, Ishida S, Nakajima H, Furutama D, Uehara H; et al. (2006). „Progression rate of ALSFRS-R at time of diagnosis predicts survival time in ALS”. Neurology. 66: 265—267..
- Armon C, Moses D. Linear estimates of rates of disease progression as predictors of survival in patients with ALS entering clinical trials. J Neurol Sci. 1998 Oct. 160 Suppl 1:S37-41.
- Armon C, Graves MC, Moses D, Forté DK, Sepulveda L, Darby SM, et al. „Linear estimates of disease progression predict survival in patients with amyotrophic lateral sclerosis”. Muscle Nerve. 23 (6): 874—82. јун 2000..
- Armon C, Brandstater ME (новембар 1999). „Motor unit number estimate-based rates of progression of ALS predict patient survival”. Muscle Nerve. 22 (11): 1571—5..
- Czaplinski A, Yen AA, Appel SH (новембар 2006). „Amyotrophic lateral sclerosis: early predictors of prolonged survival”. J Neurol. 253 (11): 1428—36..
- Roche JC, Rojas-Garcia R, Scott KM, Scotton W, Ellis CE, Burman R, et al. A proposed staging system for amyotrophic lateral sclerosis (март 2012). Brain. 135: 847—52. Недостаје или је празан параметар
|title=(помоћ). - Kim WK, Liu X, Sandner J, Pasmantier M, Andrews J, Rowland LP; et al. (2009-11-17). „Study of 962 patients indicates progressive muscular atrophy is a form of ALS”. Neurology. 73 (20): 1686—92..
- Armon C, Schultz JD. Preferences of patients with ALS for accurate prognostic information. Presented at the annual meeting of the International Alliance of Motor Neuron Disease Associations. Philadelphia. December 2004.
- Armon C. ALS 1996 and Beyond: New Hopes and Challenges. A Manual for Patients, Families and Friends. Loma Linda University Department of Neurology, Loma Linda, California. Fourth Edition. 2007.
- Isaacs JD, Dean AF, Shaw CE, Al-Chalabi A, Mills KR, Leigh PN (јул 2007). „Amyotrophic lateral sclerosis with sensory neuropathy: part of a multisystem disorder?”. J Neurol Neurosurg Psychiatry. 78 (7): 750—3..
- Byrne S, Elamin M, Bede P, Hardiman O (април 2012). „Absence of consensus in diagnostic criteria for familial neurodegenerative diseases”. J Neurol Neurosurg Psychiatry. 83 (4): 365—7..
- Byrne S, Bede P, Elamin M, Kenna K, Lynch C, McLaughlin R; et al. (мај 2011). „Proposed criteria for familial amyotrophic lateral sclerosis”. Amyotroph Lateral Scler. 12 (3): 157—9..
- de Carvalho M, Dengler R, Eisen A; et al. (март 2008). „Electrodiagnostic criteria for diagnosis of ALS”. Clin Neurophysiol. 119 (3): 497—503..
- Bromberg MB, Swoboda KJ, Lawson VH. Counting motor units in chronic motor neuropathies. Exp Neurol. 2003 Nov. 184 Suppl 1:S53-7. *
Armon C, Brandstater ME. „Motor unit number estimate-based rates of progression of ALS predict patient survival”. Muscle Nerve. 22 (11): 1571—5. новембар 1999..
- Cedarbaum JM, Stambler N. Performance of the Amyotrophic Lateral Sclerosis Functional Rating Scale (ALSFRS) in multicenter clinical trials. J Neurol Sci. 1997 Oct. 152 Suppl 1:S1-9.
- Cedarbaum JM, Stambler N, Malta E, et al. The ALSFRS-R: a revised ALS functional rating scale that incorporates assessments of respiratory function. BDNF ALS Study Group (Phase III). J Neurol Sci. 1999 Oct 31. 169(1-2):13-21.
- Instructions for completing the ALSFRS-R. (ALS Functional Rating Scale). ALS Connection.
- Miller RG, Rosenberg JA, Gelinas DF, et al. Practice parameter: the care of the patient with amyotrophic lateral sclerosis (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology: ALS Practice Parameters Task Force. Neurology. 52 (7): 1311—23. 1999-04-22. Недостаје или је празан параметар
|title=(помоћ). - Bedlack RS, Mitsumoto H, Eds. Amyotrophic Lateral Sclerosis: A Patient Care Guide for Clinicians. New York: Demos Medical Publishing; 2012.
- Miller RG, Mitchell JD, Lyon M, Moore DH. Riluzole for amyotrophic lateral sclerosis (ALS)/motor neuron disease (MND). Cochrane Database Syst Rev. 2007 Jan 24. CD001447.
- Bensimon G, Lacomblez L, Delumeau JC, Bejuit R, Truffinet P, Meininger V (мај 2002). „A study of riluzole in the treatment of advanced stage or elderly patients with amyotrophic lateral sclerosis”. J Neurol. 249 (5): 609—15..
- Yanagisawa N, Tashiro K, Tohgi H,; et al. (1997). Igakuno Ayumi. 182: 851—866. Недостаје или је празан параметар
|title=(помоћ). [Article in Japanese]. - Yanagisawa N, Shindo M. [Neuroprotective therapy for amyotrophic lateral sclerosis (ALS)]. Rinsho Shinkeigaku. 36 (12): 1329—30. децембар 1996. Недостаје или је празан параметар
|title=(помоћ). - Armon C, Guiloff RJ, Bedlack R (септембар 2002). „Limitations of inferences from observational databases in amyotrophic lateral sclerosis: all that glitters is not gold”. Amyotroph Lateral Scler Other Motor Neuron Disord. 3 (3): 109—12..
- Bourke SC, Tomlinson M, Williams TL, Bullock RE, Shaw PJ, Gibson GJ (2006). Lancet Neurol. 5: 140—147. Недостаје или је празан параметар
|title=(помоћ). [Medline]. - Jackson CE, Gronseth G, Rosenfeld J; et al. (фебруар 2009). „Randomized double-blind study of botulinum toxin type B for sialorrhea in ALS patients”. Muscle Nerve. 39 (2): 137—43..
- Neppelberg E, Haugen DF, Thorsen L, Tysnes OB (децембар 2007). „Radiotherapy reduces sialorrhea in amyotrophic lateral sclerosis”. Eur J Neurol. 14 (12): 1373—7.. [Medline].
- Brooks BR, Thisted RA, Appel SH, et al. Treatment of pseudobulbar affect in ALS with dextromethorphan/quinidine: a randomized trial. Neurology. 63 (8): 1364—70. 2004-10-26. Недостаје или је празан параметар
|title=(помоћ). - Pioro EP, Brooks BR, Cummings J, Schiffer R, Thisted RA, Wynn D (новембар 2010). „Dextromethorphan plus ultra low-dose quinidine reduces pseudobulbar affect”. Ann Neurol. 68 (5): 693—702..
- Baile WF, Buckman R, Lenzi R, Glober G, Beale EA, Kudelka AP (2000). „SPIKES-A six-step protocol for delivering bad news: application to the patient with cancer”. Oncologist. 5 (4): 302—11..
- VA Secretary Establishes ALS as a Presumptive Compensable Illness. September 23, 2008. VA Secretary Establishes ALS as a Presumptive Compensable Illness. September 23, 2008.
- Lo Coco D, Marchese S, Pesco MC, La Bella V, Piccoli F, Lo Coco A (2006-09-12). „Noninvasive positive-pressure ventilation in ALS: predictors of tolerance and survival”. Neurology. 67 (5): 761—5..
- Harding A. PET scans distinguish ALS patients from healthy controls. Reuters Health Information. March 11, 2014.
- Migliore L, Coppede F. Genetics, environmental factors and the emerging role of epigenetics in neurodegenerative diseases. Mutat Res. 2008 Oct 31.
- Valdmanis PN, Rouleau GA (2008-01-08). „Genetics of familial amyotrophic lateral sclerosis”. Neurology. 70 (2): 144—52..
- Van Laere K, Vanhee A, Verschueren J, et al. Value of 18fluorodeoxyglucose-positron-emission tomography in amyotrophic lateral sclerosis: a prospective study. JAMA Neurol. 2014 Mar 10.
- Brooks BR. El Escorial World Federation of Neurology criteria for the diagnosis of amyotrophic lateral sclerosis. Subcommittee on Motor Neuron Diseases/Amyotrophic Lateral Sclerosis of the World Federation of Neurology Research Group on Neuromuscular Diseases and the El Escorial "Clinical limits of amyotrophic lateral sclerosis" workshop contributors. J Neurol Sci. 1994 Jul. 124 Suppl:96-107.
- de Carvalho M, Dengler R, Eisen A, England JD, Kaji R, Kimura J; et al. (март 2008). „Electrodiagnostic criteria for diagnosis of ALS”. Clin Neurophysiol. 119 (3): 497—503..
- Chiò A, Battistini S, Calvo A, Caponnetto C, Conforti FL, Corbo M, et al. „Genetic counselling in ALS: facts, uncertainties and clinical suggestions”. J Neurol Neurosurg Psychiatry. 85 (5): 478—85. мај 2014..
- Zu T, Gibbens B, Doty NS, Gomes-Pereira M, Huguet A, Stone MD; et al. (2011-01-04). „Non-ATG-initiated translation directed by microsatellite expansions”. Proc Natl Acad Sci U S A. 108 (1): 260—5..
- Zu T, Liu Y, Bañez-Coronel M, Reid T, Pletnikova O, Lewis J, et al. RAN proteins and RNA foci from antisense transcripts in C9ORF72 ALS and frontotemporal dementia. Proc Natl Acad Sci U S A. 2013 Dec 17. 110 (51):E4968-77.
- Al-Chalabi A, Calvo A, Chio A, Colville S, Ellis CM; et al. (новембар 2014). „Analysis of amyotrophic lateral sclerosis as a multistep process: a population-based modelling study”. Lancet Neurol. 13 (11): 1108—13..
- Fournier CN, Gearing M, Upadhyayula SR, Klein M, Glass JD (2015-04-28). „Head injury does not alter disease progression or neuropathologic outcomes in ALS”. Neurology. 84 (17): 1788—95..
- Woolley SC, Strong MJ (новембар 2015). „Frontotemporal Dysfunction and Dementia in Amyotrophic Lateral Sclerosis”. Neurol Clin. 33 (4): 787—805..
Spoljašnje veze[уреди | уреди извор]
| Klasifikacija | |
|---|---|
| Spoljašnji resursi |
![]() Медији везани за чланак Amiotrofična lateralna skleroza на Викимедијиној остави
Медији везани за чланак Amiotrofična lateralna skleroza на Викимедијиној остави
- Les Turner ALS Fondacija
- Dodatna pomoć za ALS
- ALS Asocijacija
- Stephen Hawking - vebsajt
- Klinička ispitivanja Neurodeksa
- Lou Gehrig - vebsajt Архивирано на сајту Wayback Machine (9. мај 2008)
- ALS-Projekat - Besplatni komunikacioni softver za ALS bolesnike
- Fondacija za razvoj ALS terapije
- PatientsLikeMe
- Duke ALS Klinika
- Robert Packard centar za ALS istraživanje
- Grupa za podršku ALS bolesnika
- ALS-TDF forumi
- BrainTalk forum
- BUILD UK
- Patrick O'Brien fondacija
 | Molimo Vas, obratite pažnju na važno upozorenje u vezi sa temama iz oblasti medicine (zdravlja). |
