SIB-1757
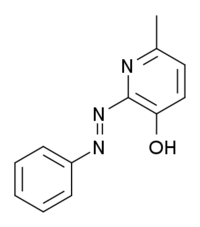 | |
| IUPAC ime | |
|---|---|
6-metil-2-(fenilazo)-3-piridinol | |
| Identifikatori | |
| CAS broj | 31993-01-8 |
| ATC kod | none |
| PubChem | CID 6849066 |
| Hemijski podaci | |
| Formula | C12H11N3O |
| Molarna masa | 213,235 g/mol |
| |
SIB-1757 je lek koji se koristi u naučnim istraživanjima. On je bio jedan od prvih jedinjenja razvijenih kao selektivni antagonisti za metabotropni glutamatni receptor podtip mGluR5.[1] On ima antihipergleziono dejstvo kod životinja.[2] SIB-1757 zajedno sa drugim mGluR5 antagonistima ima neuroprotektivne i hepatoprotektivne efekte,[3][4] i on se takođe koristi u studiranju uloge mGluR5 receptora tokom moždanog razvoja.[5]
Reference[уреди | уреди извор]
- ^ Varney MA, Cosford ND, Jachec C, Rao SP, Sacaan A, Lin FF, Bleicher L, Santori EM, Flor PJ, Allgeier H, Gasparini F, Kuhn R, Hess SD, Veliçelebi G, Johnson EC (1999). „SIB-1757 and SIB-1893: selective, noncompetitive antagonists of metabotropic glutamate receptor type 5”. The Journal of Pharmacology and Experimental Therapeutics. 290 (1): 170—81. PMID 10381773.
- ^ Dogrul A, Ossipov MH, Lai J, Malan TP, Porreca F (2000). „Peripheral and spinal antihyperalgesic activity of SIB-1757, a metabotropic glutamate receptor (mGLUR(5)) antagonist, in experimental neuropathic pain in rats”. Neuroscience Letters. 292 (2): 115—8. PMID 10998562. doi:10.1016/S0304-3940(00)01458-0.
- ^ Storto M, Ngomba RT, Battaglia G, Freitas I, Griffini P, Richelmi P, Nicoletti F, Vairetti M (2003). „Selective blockade of mGlu5 metabotropic glutamate receptors is protective against acetaminophen hepatotoxicity in mice”. Journal of Hepatology. 38 (2): 179—87. PMID 12547406. doi:10.1016/S0168-8278(02)00384-7.
- ^ Fazal A, Parker F, Palmer AM, Croucher MJ (2003). „Characterisation of the actions of group I metabotropic glutamate receptor subtype selective ligands on excitatory amino acid release and sodium-dependent re-uptake in rat cerebrocortical minislices”. Journal of Neurochemistry. 86 (6): 1346—58. PMID 12950444. doi:10.1046/j.1471-4159.2003.01932.x.
- ^ Bonsi P, Cuomo D, De Persis C, Centonze D, Bernardi G, Calabresi P, Pisani A (2005). „Modulatory action of metabotropic glutamate receptor (mGluR) 5 on mGluR1 function in striatal cholinergic interneurons”. Neuropharmacology. 49. Suppl 1: 104—13. PMID 16005029. doi:10.1016/j.neuropharm.2005.05.012.
