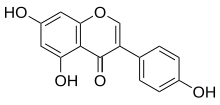
Genistein
| |

| |
| Nazivi | |
|---|---|
| IUPAC naziv
5,7-Dihidrokdi-3-(4-hidrokdifenil)hromen-4-on
| |
| Drugi nazivi
4',5,7-Trihidroksiizoflavon
| |
| Identifikacija | |
3D model (Jmol)
|
|
| ChEBI | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.006.524 |
| KEGG[1] | |
| UNII | |
| |
| Svojstva | |
| C15H10O5 | |
| Molarna masa | 270,24 g·mol−1 |
Ukoliko nije drugačije napomenuto, podaci se odnose na standardno stanje materijala (na 25 °C [77 °F], 100 kPa). | |
| Reference infokutije | |
Genistein je fitoestrogen koji pripada kategoriji izoflavona. Genistein je prvi put izolovan 1899 iz Genista tinctoria, po čemu je dobio ime. Struktura jedinjenja je određena 1926, kad je utvrđeno da je identična sa prunetolom. Prvi put je sintetisan 1928.[4]
Rasprostranjenost u prirodi[уреди | уреди извор]
Izoflavoni poput genisteina i dajidzeina su prisutni u brojnim biljkama koje se koriste u ishrani,[5][6] kao i u lekovitom bilju, Flemingia vestita[7] i Flemingia macrophylla,[8][9] i u kafi.[10] Takođe je prisutan u Maackia amurensis ćelijskim kulturama.[11]
Reference[уреди | уреди извор]
- ^ Joanne Wixon; Douglas Kell (2000). „Website Review: The Kyoto Encyclopedia of Genes and Genomes — KEGG”. Yeast. 17 (1): 48—55. doi:10.1002/(SICI)1097-0061(200004)17:1<48::AID-YEA2>3.0.CO;2-H.
- ^ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today. 15 (23-24): 1052—7. PMID 20970519. doi:10.1016/j.drudis.2010.10.003.
- ^ Evan E. Bolton; Yanli Wang; Paul A. Thiessen; Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry. 4: 217—241. doi:10.1016/S1574-1400(08)00012-1.
- ^ Walter ED (1941). „Genistin (an isoflavone glucoside) and its aglucone, genistein, from soybeans”. J Am Chem Soc. 62 (12): 3273—3276. doi:10.1021/ja01857a013.
- ^ Coward L, Barnes NC, Setchell KD, Barnes S (1993). „Genistein, daidzein, and their β-glycoside conjugates: antitumor isoflavones in soybean foods from American and Asian diets”. J Agric Food Chem. 41 (11): 1961—1967. doi:10.1021/jf00035a027.
- ^ Kaufman PB, Duke JA, Brielmann H, Boik J, Hoyt JE (1997). „A comparative survey of leguminous plants as sources of the isoflavones, genistein and daidzein: implications for human nutrition and health.”. J Altern Complement Med. 3 (1): 7—12. PMID 9395689. doi:10.1089/acm.1997.3.7.
- ^ Rao, HSP; Reddy, KS (1991). „Isofavones from Flemingia vestita”. Fitoterapia. 62 (5): 458.
- ^ Li BQ, Song QS (2009). „Chemical constituents in roots of Flemingia macrophylla”. Chinese Traditional and Herbal Drugs. 40 (2): epub. doi:10.1016/S0031-9422(00)80163-6. Архивирано из оригинала 29. 10. 2013. г. Приступљено 29. 09. 2013.
- ^ Wang BS, Juang LJ, Yang JJ, Chen LY, Tai HM, Huan MH (2012). „Antioxidant and antityrosinase activity of Flemingia macrophylla and Glycine tomentella roots”. Evidence-Based Complementary and Alternative Medicine: 1—7. PMID 22997529. doi:10.1155/2012/431081. 431081.
- ^ Alves RC, Almeida IM, Casal S, Oliveira MB (2010). „Isoflavones in coffee: influence of species, roast degree, and brewing method.”. J Agric Food Chem. 58 (5): 3002—3007. PMID 20131840. doi:10.1021/jf9039205.
- ^ Fedoreyev SA, Pokushalov TV, Veselova MV, Glebko LI, Kulesh NI, Muzarok TI, Seletskaya LD, Bulgakov VP, Zhuravlev YN (2000). „Isoflavonoid production by callus cultures of Maackia amurensis” (PDF). Fitoterapia. 71 (4): 365—72. PMID 10925005. doi:10.1016/S0367-326X(00)00129-5. Архивирано из оригинала (PDF) 22. 05. 2013. г. Приступљено 29. 09. 2013.
Spoljašnje veze[уреди | уреди извор]
- Compound Summary at NCBI PubChem
- Information at Drugfuture
- Fact Sheet at Zerobreastcancer Архивирано на сајту Wayback Machine (21. април 2021)
